Overcoming nicotine addiction often requires repeated provider interventions and multiple patient attempts.
The use of FDA-approved medications helps reduce withdrawal symptoms and increases your patients’ smoking cessation success.
Evidence shows combining long-acting + short-acting nicotine replacement therapy (NRT) can more than triple long-term quit rates.
FDA-approved medication and cessation counseling are effective when used by themselves for treating tobacco dependence. However, the combination of medication and counseling is more effective than either alone.
NYS Medicaid covers evidence-based smoking cessation treatment, including all seven FDA-approved medications when prescribed. Private health insurance and Medicare plans vary.
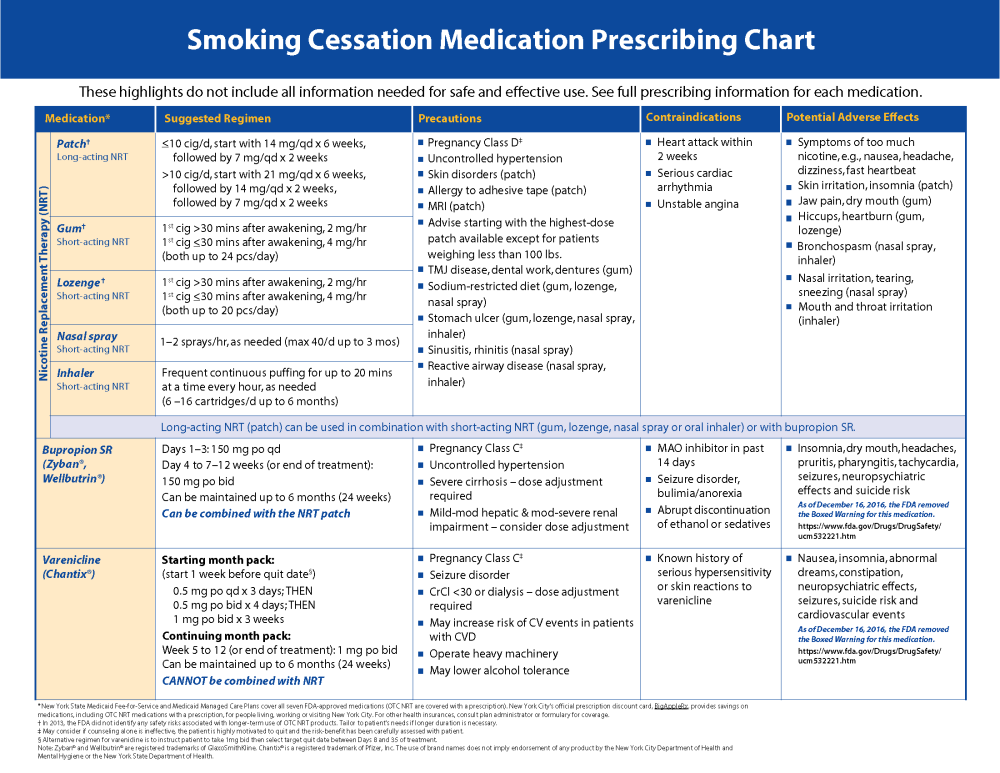
The U.S. Food and Drug Administration (FDA) has approved:
- Four nicotine replacement therapies (NRT):
- transdermal patch*
- gum*
- lozenge*
- nasal spray†
- Two non-nicotine medications:
- varenicline
- bupropion
* available over-the-counter (OTC) and covered by Medicaid with a fiscal order and some health insurance plans with prescription
†requires prescription
Combination Pharmacotherapy
Not all smokers can quit with single-drug therapy. Evidence exists that combination pharmacotherapy increases long-term abstinence rates relative to placebo treatments. Note: Varenicline should not be combined with any other smoking cessation medication.
- Combination long-acting + short-acting NRT:
- provides a stable baseline nicotine level from the sustained release transdermal patch with immediate release nicotine gum, lozenge, nasal spray or oral inhaler;
- may provide a therapeutic advantage by increasing nicotine concentrations to help prevent the onset of severe withdrawal symptoms;
- may be particularly effective in suppressing tobacco withdrawal symptoms and especially useful for highly dependent smokers or those with a history of severe withdrawal.
- Bupropion + long-acting transdermal patch:
- Monitored treatment with bupropion and an NRT patch is more effective in increasing long-term cessation abstinence than either medication alone.
Medicaid and FDA changes make it easier to prescribe safe and effective smoking cessation regimens for your patients:
- Medicaid:
- removed the two-course annual limit for smoking cessation medications, including combination pharmacotherapy; and
- removed prior authorization for prescribing cessation medication (except for brand-name products when generics are available).
- FDA:
- determined there are no significant safety concerns with prescribing combination nicotine replacement therapy (NRT) or using NRT even if a person is still smoking;
- determined it is safe in most cases to use NRT longer than the 8 to 12 weeks that label directions may state; and
- evaluated varenicline and bupropion and found the benefits outweigh the risks of using them for most people. As a result, FDA approved the removal of the boxed warning about potential neuropsychiatric side effects from the drug labels of both Chantix and Zyban when used for smoking cessation.
Are E-Cigarettes a Quit Smoking Aid?
E-cigarettes are not an FDA-approved smoking cessation medication. The FDA classifies e-cigarettes as a tobacco product, just like the cigarettes, cigars, smokeless tobacco, and other tobacco products you advise and assist your patients in quitting.
The seven FDA-approved smoking cessation products are regulated by the FDA Center for Drug Evaluation and Research (CDER). New drugs must be evaluated and approved by the FDA before they can be sold and marketed in the U.S. This process aims to ensure that over-the-counter and prescription drugs are safe and effective and that their benefits outweigh any known associated risks.
E-cigarettes, on the other hand, are regulated by the FDA Center for Tobacco Products (CTP). Although the FDA has had broad regulatory authority over e‐cigarettes since 2016, it currently conducts little oversight of the products. In addition, unlike combustible cigarettes, there is no unit standardization for the devices or of the nicotine content and other ingredients contained in e-cigarette liquids (e-liquids).
In 2018, the National Academies of Sciences, Engineering, and Medicine (NASEM) published a report, Public Health Consequences of E-Cigarettes. NASEM found conclusive evidence that completely substituting e-cigarettes for combustible tobacco cigarettes reduces users' exposure to numerous toxicants and carcinogens present in combustible tobacco cigarettes. However, NASEM also concluded there is insufficient evidence from randomized controlled trials about the effectiveness of e-cigarettes as cessation aids compared with no treatment or to FDA–approved smoking cessation treatments.
The U.S. Preventive Services Task Force (USPSTF) concludes that the current evidence is insufficient to recommend e-cigarettes for tobacco cessation in adults, including pregnant women. The USPSTF recommends that clinicians provide patients who use tobacco with the effective and safe cessation interventions identified in the U.S. Public Health Service Treating Tobacco Use and Dependence: 2008 Update—Clinical Practice Guidelines.
Research is underway to evaluate what utility, if any, e-cigarettes may have in helping people quit smoking. Should evidence support the use of e-cigarettes in cessation, manufacturers must submit the product to the FDA as a new drug to be evaluated, and the e-cigarette must be FDA-approved before it can be marketed and sold as a pharmaceutical for smoking cessation.

For More Information
- Update on the Smoking Cessation Benefit in Medicaid Fee-for-Service and Medicaid Managed Care (March 2017) – New York State Department of Health
- FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings (December 2016) – U.S. Food and Drug Administration
- Treating Tobacco Use and Dependence: 2008 Update (U.S. Department of Health and Human Services, Public Health Service)
- FDA 101: Smoking Cessation Products (August 2017) – U.S. Food and Drug Administration
- Combination Nicotine Replacement Therapy (NRT) (June 2016) – U.S. Department of Veterans Affairs
- Tobacco Smoking Cessation in Adults, Including Pregnant Women: Behavioral and Pharmacotherapy Interventions (December 2015) - U.S. Preventive Services Task Force
- Modifications To Labeling of Nicotine Replacement Therapy Products for Over-the-Counter Human Use (April 2, 2013) – U.S. Food and Drug Administration
Source:
Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update—Clinical Practice Guidelines. Rockville (MD): U.S. Department of Health and Human Services, Public Health Service, Agency for Healthcare Research and Quality, 2008